Make this Halloween one to remember with a truly special drop. The star is the MTL Hexacore Alien, our most refined MTL coil to date, delivering standout brightness and silky roundness. The retro-gaming collector’s box with bullet packaging also grants access to Multicore Nano Plus—an enhanced variant of the beloved Multicore MTL, tuned for higher wattages on regulated or mech setups for extra hit and flavor.
Note: all coils are wrapped clockwise.
Author: ergot_admin
For many years, the Mohawk coils were the flagship of Breakill’s Alien Lab catalog. Mohawk aliens have never been easy to balance—always highly specific in their performance—difficulties that saw them disappear from listings for a long time. Yet these very traits make them perfect for a limited edition: three SUPERSYMMETRY Limited Editions based on the famed Mohawk design.
Although the core often mirrors the gauge of the Triple‑Core Symmetry line, it’s their novel triangular geometry and a fully rebalanced, tight, dense—or even “stitched”—wrap that delivers unmistakable flavor and propels compact atomizers well beyond the usual limits of a classic Alien.
As always, a coil that demands meticulous construction also requires equal care and precision in installation and upkeep: the stitched wrap must be kept clean and gently pre‑activated, and the dry‑burn interval managed in tighter steps than the coil itself might suggest. Those who love to experiment know that in this rigor lies the true pleasure of vaping.
Get ready to break the mold, and enjoy a three‑dimensional, multifaceted vaping experience!
INTRODUCTION: IN SEARCH OF TASTE
In this column we will have the pleasure of exploring some aspects that determine the aromatic performance of coils, whether they are single-wire or complex, while analyzing the technical, chemical, and physical aspects and minimizing those related to suggestions and impressions. We will therefore rely on everything that is parametric, truly measurable, and verifiable.
Trying to identify what makes a coil—whether it’s for a sub-ohm device or rebuildable—more high-performing, we can identify three virtues:
- Vapor production: That is, how much vapor a coil produces.
- Aromatic intensity and hit: These are closely linked and directly dependent on vapor production.
- Aromatic definition: That is, the ability to identify and separate all the aromatic notes. We can identify two main Aromatic “characters”, let’s call them “roundness” and “brightness”, depending on how the notes are enhanced or muted.
I don’t believe it is necessary to go into depth about what vapor production or hit means, but it is necessary to delve a bit further into the concept of flavor or aromaticity, and it is worth taking this topic from the root:
The perception of the taste and smell of certain substances is determined by the interaction of specific chemical compounds with our receptors, such as taste buds but especially olfactory cells; in fact, the senses of taste and smell are closely related.
We can immediately notice an important detail: on one hand, we have a parametric science that deals with the construction of aromas, but on the other hand there is a variable and extremely personal biological system: the phrase “to each his own” is not just a saying related to personal preferences, but biologically everyone develops a certain sensitivity to different notes, and this sensitivity is not general but specific to each particular compound.
We have seen “what” perceives tastes and odors, but not “who” is the source of them, or rather we have generically said “molecules.”
The molecules responsible for smells and flavors come in various types: aromatic compounds proper (e.g., vanillin), terpenes (such as pinene and limonene), phenols and alcohols (typically linked to floral aromas), aldehydes (cinnamon), ketones, esters, and so on… There is a true science related to the construction and reconstruction of perfumes and aromas, aptly called aromatherapy or perfumery.
In this science, fragrances are referred to in terms of “notes,” referring to the aromatic impression that develops; in particular, one speaks of top, heart, and base notes:
- Top notes are the first that are perceived. Typically they are linked to light and volatile molecules that evaporate quickly. They provide the immediate impact of a fragrance and often contribute to its sense of “freshness,” being the “edges” of our “shape.” Examples are herbal, citrus, and floral notes.
- Once the top notes have evaporated or degraded, the core of the fragrance arrives, that is the heart notes, linked to molecules that are more complex and longer lasting than the top notes. They determine the main character and personality of a fragrance, acting as a bridge between the top and base notes. Typically, these are spicy or fruity notes.
- Finally, there are the base notes, or tail notes, which are the final phase of an aroma. They are usually rich, heavy molecules with low volatility, meaning they evaporate slowly. The base notes provide depth and warmth and anchor the entire composition—typically woody or spicy notes, as well as vanilla.
HEAT, REACTIVITY, AND AROMATIC SEPARATION
Why is all of this important and how does it affect what happens in our atomizers?
In our atomizers, we heat our flavored liquid at a generally controlled temperature: heat influences the expression of the different aromatic notes and the overall outcome of the experience.
Heat increases the evaporation rate of aromatic molecules—that is, it increases vapor production; however, excessive heat or heat reached too rapidly can also cause too-rapid dissipation of some delicate top notes, altering the balance. Similarly, the heart notes also prefer moderate heat to express maximum complexity, while the base notes are more resistant and suitable for higher temperatures.
We have now reached the crux of the matter: we have the tools to understand WHAT to look for in a “perfect” coil—the maximum applicable power, with the utmost respect for all the notes.
It becomes evident that if a coil is too reactive and reaches excessively high temperatures too quickly, it inhibits the expression of the top notes and some heart notes, transforming a complex liquid into an unformed, sweet paste.
Conversely, a coil that stays too cool might not sufficiently express the base notes.
The ideal strategy is therefore a gradual heating, or ramp-up, that reaches a consistent temperature.
We have understood that the ramp-up is crucial to preserving all the notes; we have “equalized” our aromatic Hi-Fi in the best possible way, and now we want… volume!
Increasing vapor production is probably the first parameter sought, a source of great satisfaction. The first thing a new vaper asks is: how can I make more vapor?
To achieve this, one might easily think that it is enough to increase the power, but if we simply increase the power we would only raise the heat—we also need to increase the evaporation surface.
Obtaining a greater surface while maintaining the thermal balance necessary to express all the aromatic notes requires that this surface be large enough to handle the provided power and therefore… it is time to leave the realm of chemistry and delve a bit into the physics of coils!
BASICS OF THERMODYNAMICS: THE JOULE EFFECT AND HEAT FLUX
From the very first day we started vaping, we were taught about Ohm’s law, which is very important for understanding the relationship between current intensity, voltage, and ohms; although crucial, it is really useless on its own in determining the efficiency and effectiveness of a coil. Imagine a one-ohm coil intended to vape at 14 watts—everything seems fine, right?
But if it is made with 20 wraps on a 4mm ID with a 20 AWG wire… it will never work! Why?
Because the mass is too high for the power expressed and required by our system!
We are about to enter the rabbit hole, and if what I have said above seems obvious, you will realize how often this concept is not applied:
To fully understand the problem, we must use another tool: Joule’s law and the Joule effect:
Q = I² × R × t
Where:
- Q represents the heat produced, measured in joules (J).
- I represents the current flowing through the conductor, measured in amperes (A).
- R represents the resistance of the conductor, measured in ohms (Ω).
- t represents the time duration during which the current flows, measured in seconds (s).
This equation represents the calculation of the heat generated (in joules) when an electric current (in amperes) flows through a conductor with a certain resistance (in ohms) for a specific duration (in seconds).
You will notice that mass does not appear in this equation, but it is necessary to understand what happens if, for the same amount of heat—that is, the same number of joules produced—this heat is applied to a higher or lower mass.
Determining this relationship is the heat flux, or the heat flow, which is the formula that relates the heat produced to the surface area over which it is applied. Mathematically, the heat flux is defined by the letter q and measured in watts per square meter:
q = Q / (A ⋅ t)
Where:
- Q is the amount of transferred heat, measured in joules (J).
- A is the surface area through which the heat transfer takes place, measured in square meters.
- t is the time duration during which the heat transfer occurs, measured in seconds (s).
It is generally accepted that a good heat flux is between 150 and 300 mW/mm², but of course, this is subjective and does not take into account the final and even more important factors: the amount of air entering the system to moderate the temperature and the liquid’s role in moderating the temperature.
For now, we are interested in finding the relationship between the coil’s surface area and the applied power: I will spare you the trivial mathematical steps; we combine Joule’s law, Ohm’s law, and heat flux to obtain:
q = P / A
Looking at the formula once again, we notice an interesting point: for the purpose of the amount of heat, ohms (in a regulated system) are not relevant to the system’s thermodynamics! What matters is the surface area and the applied power! Of course, if you enter the world of mechanical mods, the ohm value must be considered to obtain the desired power, but it does not change the crux of the matter—namely, that if a coil in our possession appears not energetic and reactive enough for our tastes, or if it has too much surface area, regardless of its ohm value, it is necessary either to reduce the mass or to apply higher power.
In summary, an ideal coil should be reactive but gradual, reaching the ideal temperature for all the notes of the aromatic bouquet. Now we know that to achieve this, a precise balance between surface area and applied power is necessary.
The formula obtained allows us to understand that to increase vapor production while respecting all the notes, it is necessary to increase the surface area, maintaining a balance with the wattage. We do not actually need a coil with a lower ohm, but one with a greater surface area! Obviously, the two things are linked, and factors such as battery stress and their lifespan come into play, but that is not the topic for today.
THE IMPACT OF COMPLEX COILS ON AROMATIC PERFORMANCE
We have reached the end of our banquet—it’s dessert time.
At a certain point in the evolution of rebuildable devices, once cloud chasing became popular, people tried to achieve ever more surface area. Flat wires were born, and although they increased the surface in terms of width, in absolute terms—let’s say by mass—it was difficult to achieve a balance because the coils were too wide and heated up too quickly.
Thus, parallel coils were developed, which, by exploiting parallel resistance, achieved a better balance between surface area and applied power. People started twisting wires together—first two, then three—and something very interesting was discovered: when twisting the wire, the performance was much better. Why?
Here enters our friend capillarity: twisting the wires together creates channels that distribute the liquid not only on one side of the resistance but also within and along the entire path, constantly moderating the temperature. Experiments continued by the well-known figure blueeyedgoon85, creator of the first true complex coils—clapton, fused clapton, alien, staggered, etc.
What happens in these coils? An internal capillarity is created within the resistance, whose role—its ability to preserve and express the entire aromatic bouquet we discussed earlier—is essentially linked to its capacity to constantly moderate the temperature and to increase the evaporation surface, expanding it from the two initial dimensions of the single central channel of the single-wire resistance to the three dimensions of all the internal microchannels created by complex wrap and core designs.
At this point, another factor completely changes the rules of the game, namely having a wrap, which, while not altering the resistance, does add mass: the balance between surface area and wattage must therefore be recalculated.
What does this mean? It means that depending on the type of wrap and its dimensions, we will obtain different capillarity and surface area.
The balance between the thickness of the wrap and the core is crucial; the thinner it is, the easier it is to obtain high-performing coils that, without the need for excessive power, can reach the ideal temperature.
Even the total number of wraps becomes important—they must be sufficient to balance the speed of heating with the applied power and, if you’re in a mechanical setup, also with the ohm value necessary to achieve the desired wattage.
Before delving into the world of custom coils and concluding this exploration, I would like to briefly analyze why mesh coils work so well on “pod” devices and so poorly on rebuildable atomizers.
The attempt to exploit the capillarity of the mesh clashes with an objectively small surface area, which heats up at an incredible rate, therefore being unsuitable for high power but extraordinarily efficient at reduced power. However, the aromatic intensity, the separation of notes, and the hit are not increased, because to achieve that it is necessary to increase the power while accompanying it with maximum surface area and airflow.
Here, complex coils come into play—a completely different strategy to achieve maximum aromatic efficiency (rather than energetic efficiency).
Let’s briefly analyze, point by point, summarizing in a few words: what is the difference between a clapton, a fused, an alien, and a staggered coil? Mainly, it is the amount of wrap used and the number of internal channels:
- A single clapton provides only external capillarity and little additional mass; only a few extra watts can be applied.
- A fused adds an internal channel that further moderates the temperature: the thicker the wrap, the greater the need for energy to properly activate the coil.
- An alien further increases the external capillarity, by about ⅓ compared to a fused of the same core.
- A staggered makes things even more complex, as the type of wrap provides much more capillarity but also adds more mass; therefore, the staggered approach usually performs best when the applied power becomes substantial, compensating and balancing the mass and fully exploiting the capillarity.
Thanks to what we have seen at the beginning, and having identified what determines the aromatic performance of a coil, it becomes incredibly easy to judge a coil in terms of its complexity. However, there is one last, counterintuitive factor:
It might seem at first that the greater the volume of empty space in which the capillarity is expressed, the better the performance. However, capillarity has a prerogative: it expresses itself best when it travels through more confined spaces, and his expression it’s relate on a surface area parameter, not volume!.
In fact, a 2-core fused coil has a greater “empty” volume than a staggered coil of the same dimensions (composed internally of ribbon), yet its aromatic definition is noticeably flattened out, giving a round and often too sweet output: Why?
As said, the vaporization surface does not coincide with the volume of the internal channels and capillarity distributed over very small spaces is far more efficient and provides a greater evaporation surface.
If capillarity has to work over channels that are too wide, it is not sufficiently rapid in supplying liquid to the coil’s wrap, which will then reach higher temperatures temporarily, resulting in explosive and intermittent evaporation. Typically, this results in the characteristic popping sound—not more, not less—due to the air trapped within the channels in the split seconds when it is not moderated by the liquid; as it heats up, it explodes outside the wrap, creating a depression that draws in more liquid, and so on, with a typical vaporization that greatly emphasizes the base notes but often fails to preserve the top notes, maintaining an overall high surface temperature.
That’s all for today—it has been a long journey, but I hope it has been an interesting exploration into this fantastic world!
“My atomizer X is dry/gurgling/has flooded my bag!” “Help!”… Probably the first message we all sent the first time we tried rebuilding. And as always, asking three different people will get you six different opinions, all contradicting each other: “This atomizer should be wicked like this.” “No, it should be wicked like this!” “You all know nothing! You’re all wrong! Saint Christopher appeared in my dreams and enlightened me: this is how it should be wicked!”
Alright, let’s try to provide a global solution by increasing our understanding of the system rather than relying on specific and debatable solutions. If you’ve ever had issues with your RTA not properly feeding the coil, it’s probably due to a fundamental factor many overlook: air compensation in the tank.
Wicking an RDA is straightforward, but an RTA is a different story
n RDAs, wicking is quite simple: you insert the cotton into the coil, cut it to the right length, and place it in the juice well. There are no structural limitations affecting liquid feeding, as the cotton just needs to be in contact with the soaked bottom.
In RTAs, however, the dynamics change completely. Each tank has its own structure, with wider or narrower juice flow channels and different vaporization chambers. A mistake in wicking can lead to two main problems:
- Flooding (excess liquid on the coil and gurgling)
- Dry hits (cotton not feeding properly, leading to burning and unpleasant taste)
The principle of capillarity: more cotton, more efficiency?
We have widely discussed and confirmed how a compact wick results in faster capillarity: well-packed cotton absorbs and transfers liquid to the coil more efficiently. However, this factor alone is not enough.
Excess cotton in the juice flow channels can actually hinder air rising back into the tank. If liquid is flowing down but air cannot rise, a partial vacuum forms, reducing pressure and preventing a steady liquid flow to the coil. This effect is particularly noticeable in bottom-feeding RTAs, where air compensation capacity is limited.
The solution: wick smartly
To ensure optimal feeding, you need to find the right balance:
- Ensure the cotton is well inserted into the juice flow channels but without completely blocking the passage.
- Always leave some space to allow air to rise back into the tank.
- If your RTA has very large channels, consider thinning the cotton slightly with tweezers to prevent total obstruction.
- Try different wicking lengths and observe how the liquid behaves: a good wick allows consistent feeding without flooding or dryness.
Conclusion
If your RTA isn’t wicking properly, it may not just be a capillarity issue but rather a balance between liquid and air. Make sure to wick in a way that allows proper airflow, and you might solve many feeding issues you didn’t even realize you had. Next time you rebuild your RTA, try applying this advice: you’ll notice the difference!
Have no fear! We got you covered!
Many models has been kept and converted into the new “the lab” collection!
The conversion chart will help you finding them, and find the great classics into the giant new collection!
We promised big surprises—here they are!
A collection of 31 coil models, both new and classic, combined into our most comprehensive selection ever! The new 2025 catalog revolutionizes the LAB’s offerings in every vaping style, power range, and flavor output.
Plus, all Suprema Ratio wires and spools are now 20% off forever!
DOWNLOAD THE NEW CATALOG HERE:
https://shop.breakill.com/wp-content/uploads/2024/12/CAT_ENG_25_LQ_compressed.pdf
In the world of vaping, the choice of coil material is crucial for determining performance, durability, and safety. Among the most commonly used materials are Kanthal and Nichrome, each with its own unique history and specific properties.
History and Composition
Nichrome is an alloy primarily composed of nickel and chromium, developed in the early 20th century. Thanks to its high resistivity and oxidation resistance, it quickly became a popular choice for heating elements in industrial furnaces and household appliances.
In the 1930s, engineer Hans von Kantzow introduced Kanthal, an alloy based on iron, chromium, and aluminum. This new composition offered longer durability and a higher maximum operating temperature compared to Nichrome.
Differences Between Kanthal and Nichrome
- Resistivity and Reactivity:
Nichrome has lower resistivity than Kanthal, making it more reactive and enabling faster heating. This translates to smoother output and a flavor profile often preferred by many vapers. - Maximum Operating Temperature:
Kanthal can operate at higher temperatures, up to 1425°C, compared to Nichrome’s limit of 1250°C. This makes it ideal for applications requiring extreme heat. - Durability and Stability:
Kanthal offers up to four times the lifespan of Nichrome, due to the formation of a protective aluminum oxide layer that prevents oxidation and corrosion.
Resistance to Temperature and Corrosion
Kanthal, with its aluminum content, forms an aluminum oxide layer that provides effective protection against oxidation at high temperatures. However, in humid environments or when exposed to aggressive chemicals, this layer may be less resilient.
Nichrome, on the other hand, forms a chromium oxide layer. Although this layer is less heat-resistant compared to aluminum oxide, it provides good protection in chemically aggressive environments. This makes Nichrome more suitable in scenarios where resistance to corrosion is a priority.
Passivation and Preheating
There is a widespread belief that preheating Nichrome coils is harmful. In reality, controlled preheating promotes the formation of the chromium oxide layer, a process known as passivation, which improves the material’s resistance to corrosion caused by e-liquids.
For Kanthal, passivation occurs naturally during use, forming an aluminum oxide layer. However, this layer can be less resistant in chemically aggressive environments, making Kanthal more susceptible to corrosion in such conditions.
Conclusion
The choice between Kanthal and Nichrome depends on the specific needs of the vaper. Kanthal offers greater durability and resistance to high temperatures, while Nichrome ensures superior reactivity and better corrosion resistance in chemically aggressive environments. Understanding the properties of each material and their associated passivation processes is essential to optimize the vaping experience and ensure user safety.
sources:
https://www.kanthal.com/it/knowledge-hub/inspiring-stories/unalternativa-migliore-al-nichel
All Heroes and Classic line coils are 10% off! Don’t miss it!
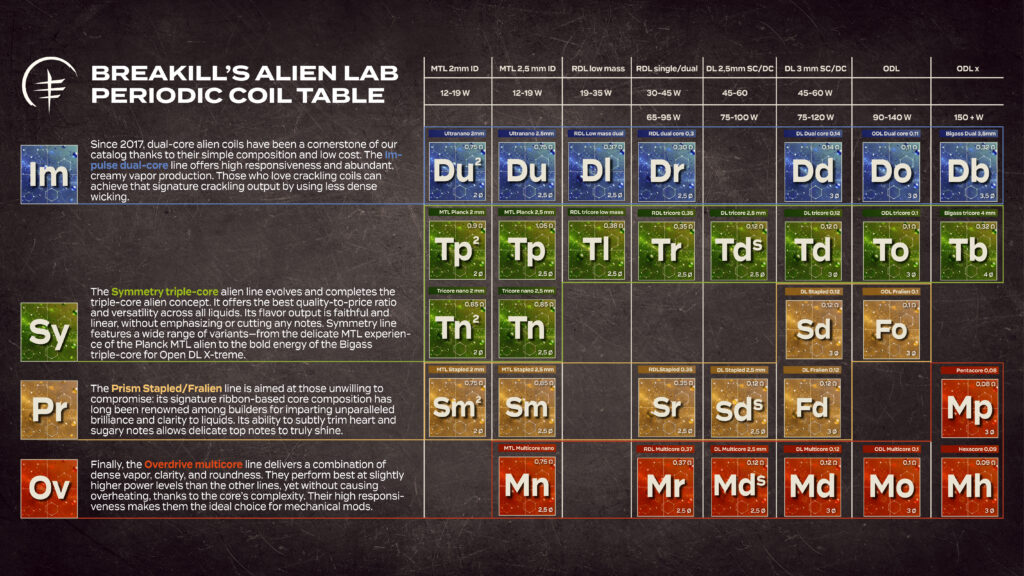
To fully understand the differences between the coils, it’s important to grasp the significance of certain parameters and terms:
Ø ID: Refers to the internal diameter of the coil in millimeters. Larger diameters typically require more energy to perform optimally but also have greater autonomy between one dry burn and the next.
Ω Ohm S.C./D.C.: The ohm value of the resistance, single coil (S.C.) and dual coil (D.C.). Typically, the ohm values of complex resistances are lower compared to simple coils and require a few more watts. This parameter should not be misunderstood: higher power results in greater vapor production and not necessarily in increased overheating of the system!
W Watt Range: The ideal power range for the coil. It’s important to stay as much as possible within the indicated ranges: you might need to apply a few more watts than usual, but this energy is applied over a larger surface area compared to a single wire, therefore it doesn’t result in more heat but in more vapor!
Dry Burn: The interval between one cleaning of the coil and the next. It can vary greatly depending on the type of liquid; it’s important to clean the coil as frequently as possible to ensure maximum longevity.
Duration: The indicative total lifespan of the coil. Linked to the dry burn interval and variable depending on the type of liquid used.
Reactivity: The speed at which the coil heats up or its ability to perform even at reduced power levels. It is usually greater in coils with smaller internal diameters or with lower ohm values.
Vapor Production: The production of vapor and aromatic intensity; it can be compared to the volume of an audio system.
Roundness: Indicates a performance that favors the blending and smoothness of aromatic notes, offering a more unified and creamy tasting experience.
Sharpness: Indicates a performance that emphasizes the separation and precision of aromatic notes, offering a more distinct gustatory experience and a drier output.
Hotspots are a phenomenon that typically occurs during coil assembly and manifests as irregular ignition, localized in small points instead of a uniform activation across the coil. Typically, on a newly installed coil, you can see spots that light up before the rest of the coil. The ohm value is also affected: because the current takes a shorter path through the resistance, the coil’s reading is much lower than expected.
An excessively low ohm value compared to expected values is always a symptom of a hotspot!
This is an almost inevitable phenomenon linked to the coil’s wrap, that is, the outer winding. It involves the creation of “alternative paths” for the current through the outer wrap instead of the inner core. It is often confused with the coil’s quality, and many myths have arisen around this phenomenon, attributed to things like wire stress (but what does that mean?) or poorly constructed coils.
The truth is that it is much more related to the coil’s complexity and the diameter of the wires used. Simply put, a finer or more complex wrap will make more windings around the core. Consequently, there will be more contact points that can create undesired paths. Similarly, more complex wraps like interlock or stitched wraps double the likelihood of hotspot formation.
Naturally, the number of wraps also matters: a big-ass coil uses twice the material compared to a “normal” coil and consequently has twice the chances of forming hotspots.
Finally, the “softness” of a coil: the more complex a coil is, the thinner the wire diameters will be, making the coil less rigid. A softer coil is more likely to come apart during assembly.
In other words, the more premium a coil is, the more experience a vaper will need to remove hotspots! Let’s look at the best solutions:
When performing the first activations or a dry burn, it’s wise to drastically reduce the wattage. Applying too much power to a hotspot could permanently weld it, making future removal impossible.
People often tend to scrape the surface to remove hotspots. While effective, this method is risky, especially on expensive coils with intricate wraps like staggerton, interlock, stitched, etc. The safest solution is to slightly separate the wraps where the hotspots form.
Prevention is better than cure! When mounting a coil on the atomizer, it’s wise to slightly separate the coil’s legs from the windings with a gentle push using a screwdriver tip. This will remove the first unwanted contact point and make hotspot removal easier.
When inserting the cotton, the coil might generate new hotspots. An ohm value reading will promptly reveal them. Fortunately, if you’ve done a good job removing them initially, they will be easy to fix—just push the wraps outward to restore the correct path.
When you remove the cotton to perform a dry burn, the coil may come apart, requiring you to remove some new hotspots. Reducing the wattage is essential to avoid damage.
A video is worth a thousand words, so here’s a quick tutorial to effectively remove hotspots: